What Is Used to Break Down Compounds in Electrolysis
The energy causes a chemical reaction and breaks the bonds holding the atoms together. They lose electrons and are oxidised.
Sodium chloride can also be broken down intoelemental sodium and chlorine gas by electrolysis.

. When the bonds are broken the compound can. This results in chemical reactions at the electrodes and the separation of materials. Compoundscan be broken apart in chemical reactions called decomposition reactions.
Compounds are those elements. For ex - electricity electrolysis. How is a compound different from the elements that break it up.
Compounds can be broken down by using heat or electrical energy. 2 n a c l s 2 n a s c l 2 g heat thermal decomposition for more info visit. Heat can also be used to break down sodium hydroxide into the simpler compounds sodium oxide and water.
Two commonly used methods of electrolysis involve molten sodium chloride and aqueous sodium chloride. For example water can be broken down intothe diatomic elementshydrogen gas and oxygen gas in a chemical reaction called electrolysis. The substance that is broken down is called the electrolyte.
How does electrolysis break compounds in to elements. Thus electrolysis finds many applications both in experimental and industrial products. Electrolysis is a chemical change produced by sending an electric current through a compound.
Yes by chemical means such as electrolysis which can break water down into oxygen and hydrogen gas. The use of an electrical current to break down compounds containing ions into their constituent elements. Water broken down to diatomic elements hydrogen gas and oxygen gas.
Electrolysis as stated above is a process of converting the ions of a compound in a liquid state into their reduced or oxidized state by passing an electric current through the compound. 2 h 2 o l 2 h 2 g o 2 g 2. What moves to the anode.
Elements and Compounds. Sodium chloride broken down to sodium chloride gas. Negatively charged ions move to the positive electrode during electrolysis.
What is the name of the substance being broken down. What moves to the cathode. Main method to break compound is decomposition.
As we have covered electrolysis is the passage of a direct electric current through an ionic substance that is either molten or dissolved in a suitable solvent.
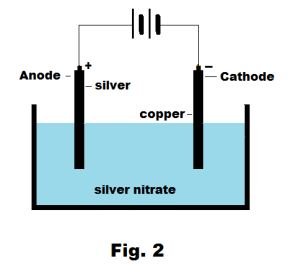
Electrochemistry Ion Electrolyte Electrolysis Electrolytic Cell Anode Cathode Ionic Compound Electroplating Faraday S Laws Of Electrolysis Electrode Potential Voltaic Cell Dry Cell Daniell Cell Gravity Cell Lead Storage Battery

New Gcse Separate Sciences Ocr Gateway Sb Page 142

How Compounds Are Broken Down Heat Electricity Video Lesson Transcript Study Com
![]()
Electrolysis Of Molten Ionic Compounds Study Com

Electrolysis And Electroplating Definition Working Principle Application

Electrolysis Aqa The Science Hive
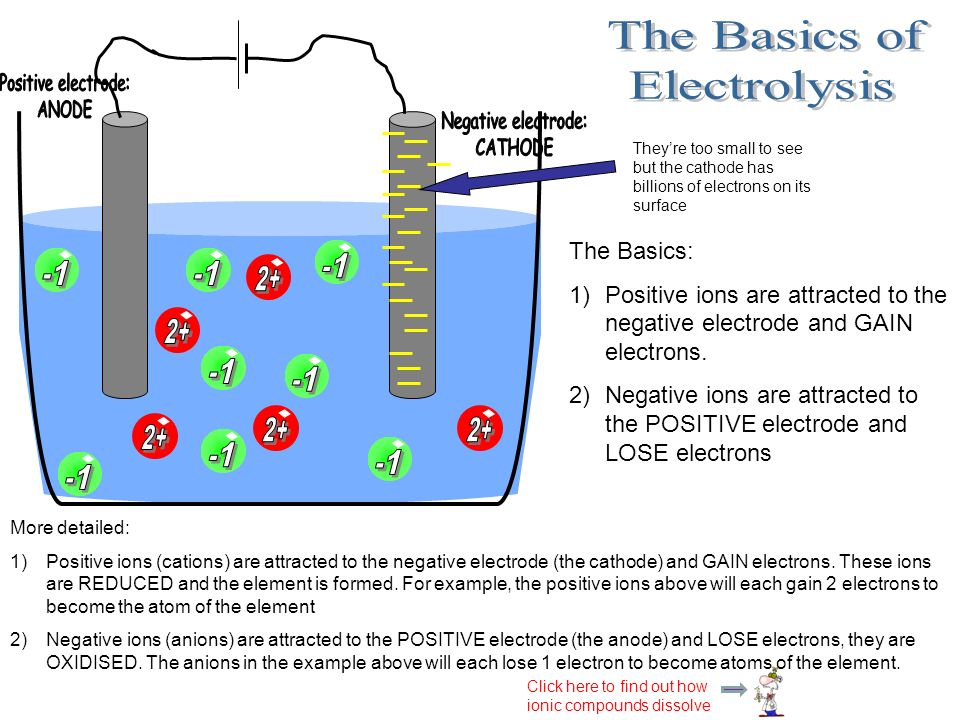
The Purpose Of Electrolysis Is To Split Up Ionic Compounds Using Electricity To Produce Useful Products Electrolysis Is Used A Lot In Industry And Is Ppt Download

The Process Of Electrolysis 4 3 1 Aqa Gcse Chemistry Revision Notes 2018 Save My Exams

7 5 Taking Compounds Apart Pg 256 Decomposition Of Compounds

Electrolysis Reaction Process What Is Electrolysis Video Lesson Transcript Study Com

Electrolysis Of Molten Lead Bromide Products Electrode Equations Anode Cathode Apparatus Electrolyte Gcse Chemistry Ks4 Science Igcse O Level Revision Notes
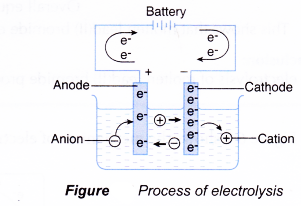
Analysing The Electrolysis Of Molten Compounds A Plus Topper

Electrolysis Of Molten Compounds 3 2 3 Edexcel Gcse Chemistry Revision Notes 2018 Save My Exams
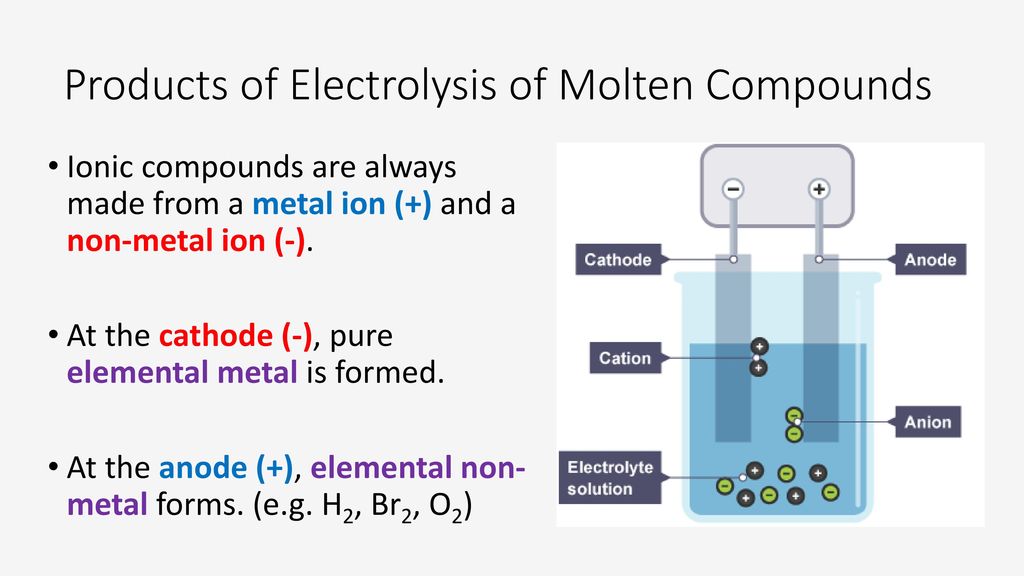
Starter Under What Conditions Do Ionic Compounds Conduct Electricity Ppt Download
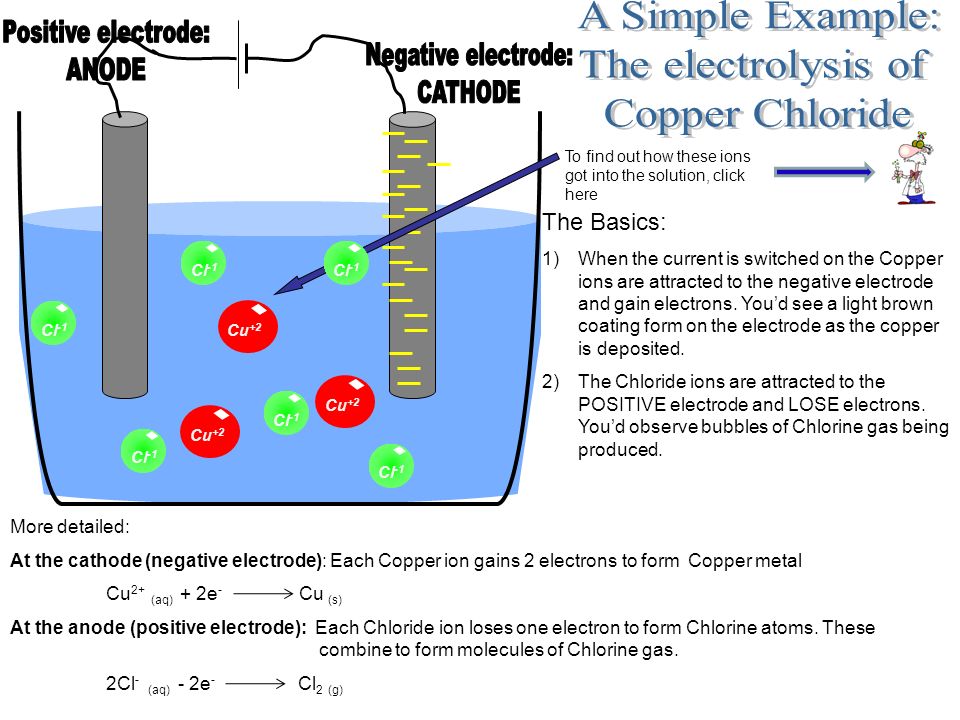
The Purpose Of Electrolysis Is To Split Up Ionic Compounds Using Electricity To Produce Useful Products Electrolysis Is Used A Lot In Industry And Is Ppt Download
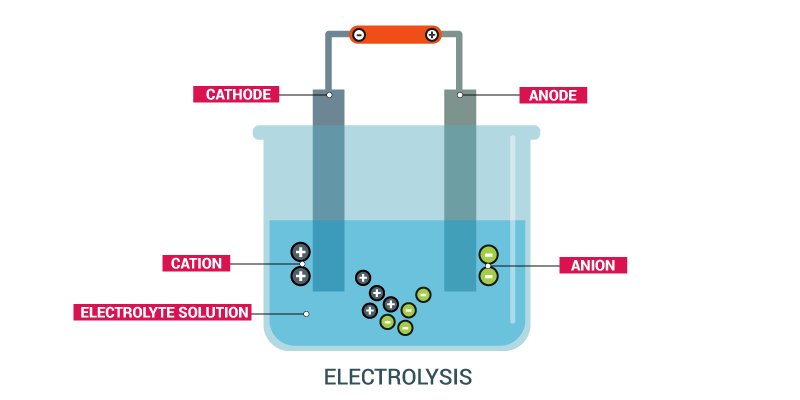
Electrolysis Of Molten Compounds Chemistry Quiz Quizizz

Electrolysis Molten Compounds 1 Molten Compounds Michael Faraday


Comments
Post a Comment